
An immune-metabolic intervention for functional HIV-cure
The unfortunate failure of two recent large prophylactic HIV-vaccine trials and a “shock and kill” functional cure approach using latency-reversing agents to eradicate the latent HIV reservoir poses a massive challenge to unraveling the immune responses that can prevent an HIV infection. Our extensive clinical studies from several cohorts on people living with HIV (PLWH) on combination antiretroviral therapy (cART) and the immune-phenotype fingerprinting of the HIV-positive elite controllers who naturally control viral replication without any therapy, indicated a profound role of immune metabolism in natural immunity to HIV-infection. Here we propose a novel approach to functional cure through immune-metabolic intervention. We perform the pre-clinical in vitro studies in novel gut/brain organ-on-chip and in vivo studies in animal models (mouse and primate) to identify the altered immunological profile due to the viral persistence or the treatment to eradicate the viral reservoir leading to a functional cure of HIV.
Publications
- Ambikan AT#, Svensson-Akusjärvi S#, Krishnan S, Sperk M, Nowak P, Vesterbacka J, Sönnerborg A, Benfeitas R, Neogi U†. Genome-scale metabolic model reveals long-term antiretroviral treatment-induced system-level metabolic shift towards oxidative phosphorylation in HIV-infection. Life Science Alliance. (accepted) [PMID: Pending]
- Svensson-Akusjärvi S, Krishnan S, Jütte BB, Ambikan AT, Gupta S, Rodriguez JE, Végvári A, Sperk M, Svensson JP, Sönnerborg A, and Neogi U† (2022) Peripheral blood CD4+CCR6+ compartment differentiates HIV-1 infected or seropositive elite controllers from long-term successfully treated individuals. Communications Biology April 13;5(1):357. doi: 10.1038/s42003-022-03315-x. [PMID: 35418589]
- Akusjärvi SS, Ambikan AT, Krishnan S, Gupta S, Sperk M, Végvári Á, Mikaeloff F, Healy K, Vesterbacka J, Nowak P, Sönnerborg A, Neogi U. Integrative proteo-transcriptomic and immunophenotyping signatures of HIV-1 elite control phenotype: A cross-talk between glycolysis and HIF signaling. iScience 2022 25;1 103607- [PMID: 35005552]
- Sperk M, Ambikan AT, Ray S, Singh K, Mikaeloff F, Diez RC, Narayanan A, Vesterbacka J, Nowak P, Sönnerborg A, Neogi U. Fecal Metabolome Signature in the HIV-1 Elite Control Phenotype: Enrichment of Dipeptides Acts as an HIV-1 Antagonist but a Prevotella Agonist. Journal of Virology 2021 95;18 e0047921- [PMID: 34232744]
- Sperk M, Mikaeloff F, Svensson-Akusjarvi S, Krishnan S, Ponnan SM, Ambikan AT, Nowak P, Sonnerborg A, Neogi U. Distinct lipid profile, low-level inflammation, and increased antioxidant defense signature in HIV-1 elite control status. iScience 2021 24;2 102111- [PMID: 33659876]
- Zhang W, Ambikan AT, Sperk M, van Domselaar R, Nowak P, Noyan K, Russom A, Sönnerborg A, Neogi U. Transcriptomics and Targeted Proteomics Analysis to Gain Insights Into the Immune-control Mechanisms of HIV-1 Infected Elite Controllers. EBioMedicine 2018 27; 40-50. [PMID: 29269040]
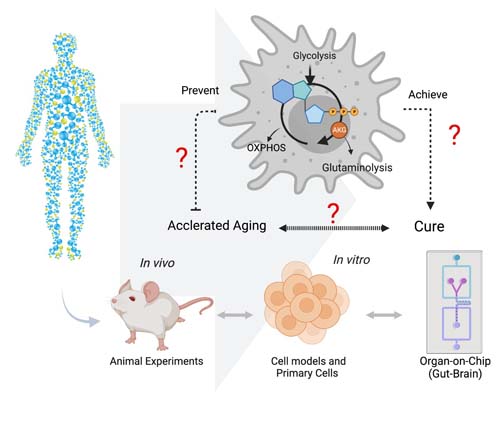
Understanding the biological mechanisms of aging
The most remarkable achievement in the battle against HIV was the discovery of effective, well-tolerated combination antiretroviral therapy (cART) which has transformed a deadly viral infection into a chronic, manageable disease. The long-term HIV infection, even with successful treatment, is associated with an accentuated onset of non-AIDS-related severe pathologies. But for reasons unknown, a significant proportion of HIV-infected individuals receiving long-term cART do not only die at an earlier age than HIV-uninfected counterparts, but they also suffer from age-related comorbidities due to chronic inflammation and immune activation during the cART. Currently, there are knowledge gaps about how HIV affects the immune system and other organs during long-term cART. We bring together system biologists, immunologists, and clinical scientists to deep immune profile long-term successfully treated PLWH in a large biobank linked European HIV aging-cohorts (COCOMO and AROGYA) to identify the PLWH at risk for age-related comorbidities and to unravel the mechanisms and cause behind. Our studies will provide clinically targetable approaches for healthy aging in PLWH.
Publications
- Mikaeloff F, Svensson Akusjärvi S, Ikomey GM, Krishnan S, Sperk M, Gupta S, Magdaleno GDV, Escós A, Lyonga E, Okomo MC, Tagne CT, Babu H, Lorson CL, Végvári Á, Banerjea AC, Kele J, Hanna LE, Singh K, de Magalhães JP, Benfeitas R, Neogi U. Trans cohort metabolic reprogramming towards glutaminolysis in long-term successfully treated HIV-infection. Communications Biology 2022 5;1 27- [PMID: 35017663]
- Olund Villumsen S, Benfeitas R, Knudsen AD, Gelpi M, Høgh J, Thomsen MT, Murray D, Ullum H, Neogi U, Nielsen SD. Integrative Lipidomics and Metabolomics for System-Level Understanding of the Metabolic Syndrome in Long-Term Treated HIV-Infected Individuals. Frontiers in Immunology 2021 12; 742736- [PMID: 35095835]
- Gelpi M, Mikaeloff F, Knudsen AD, Benfeitas R, Krishnan S, Svenssson Akusjärvi S, Høgh J, Murray DD, Ullum H, Neogi U, Nielsen SD. The central role of the glutamate metabolism in long-term antiretroviral treated HIV-infected individuals with metabolic syndrome. Aging 2021 13;19 22732-22751. [PMID: 34635603]
- Babu H, Sperk M, Ambikan AT, Rachel G, Viswanathan VK, Tripathy SP, Nowak P, Hanna LE, Neogi U. Plasma Metabolic Signature and Abnormalities in HIV-Infected Individuals on Long-Term Successful Antiretroviral Therapy. Metabolites 2019 9;10 [PMID: 31574898]

Understanding the metabolic rewiring during acute infection against RNA viruses
Natural epidemics and outbreaks of emerging and re-emerging infectious diseases are growing global problems. Events like the recent massive pandemic with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) that affected more than 187 countries, the current Crimean Congo Hemorrhagic fever (CCHF) cases in Spain, as well as dengue and Zika in Latin America and South-East Asia, reveal that the world is facing more infectious disease epidemics caused by RNA viruses. Viruses exploit the host metabolic machinery to meet their biosynthetic demands, making these host pathways potential therapeutic targets. Understanding the viral host reliance helps to pinpoint individual host genes or networks of the genes involved in the metabolic interplay of viral infection. In this project, we aim for a comprehensive mapping of metabolic perturbations during acute conditions in emerging and re-emerging RNA viruses that can aid novel antiviral strategies. After identifying the target drug, in a long-term plan, we will perform pre-clinical animal experiments.
Publications
- Ambikan AT, Yang H, Krishnan S, Svensson Akusjärvi S, Gupta S, Lourda M, Sperk M, Arif M, Zhang C, Nordqvist H, Ponnan SM, Sönnerborg A, Treutiger CJ, O’Mahony L, Mardinoglu K, Benfeitas R, Neogi U† (2021) Multiomics personalized network analyses highlight progressive immune disruption of central metabolism associated with COVID-19 severity.Cell Systems (accepted) [PMID: Pending]
- Albrich WC*, Ghosh TS*, Ahearn-Ford S, Mikaeloff F, Lunjani N, Forde B, Suh N, Kleger G, Pietsch U, Frischknecht M, Garzoni C, Forlenza R, Horgan M, Sadlier C, Negro TR, Pugin J, Wozniak H, Cerny A, Neogi U, O’Toole PW, O’Mahony L. A high-risk gut microbiota configuration associates with fatal hyperinflammatory immune and metabolic responses to SARS-CoV-2. Gut Microbes 2022 14(1):2073131. doi: 10.1080/19490976.2022.2073131.[PMID: 35574937].
- Neogi U*†, Elaldi N*, Appelberg S, Ambikan AT, Kennedy E, Bagci B, Gupta S, Rodriguez JE, Svensson-Akusjärvi S, Monteil V, Végvári A, Benfeitas R, Banerjea A, Weber F, Hewson R, Mirazimi A. Multi-omics insights into host-viral response and pathogenesis in Crimean-Congo Hemorrhagic Fever Viruses for novel therapeutic target. eLife 2022. online 19 April 2022, doi:10.7554/eLife.76071 [PMID:35437144]
- Krishnan S, Nordqvist H, Ambikan AT, Gupta S, Sperk M, Svensson-Akusjärvi S, Mikaeloff F, Benfeitas R, Saccon E, Ponnan SM, Rodriguez JE, Nikouyan N, Odeh A, Ahlén G, Asghar M, Sällberg M, Vesterbacka J, Nowak P, Végvári Á, Sönnerborg A, Treutiger CJ, Neogi U. Metabolic Perturbation Associated With COVID-19 Disease Severity and SARS-CoV-2 Replication. Molecular & Cellular Proteomics : MCP 2021 20; 100159- [PMID: 34619366]
- 5. Appelberg S, Gupta S, Svensson Akusjärvi S, Ambikan AT, Mikaeloff F, Saccon E, Végvári Á, Benfeitas R, Sperk M, Ståhlberg M, Krishnan S, Singh K, Penninger JM, Mirazimi A, Neogi U. Dysregulation in Akt/mTOR/HIF-1 signaling identified by proteo-transcriptomics of SARS-CoV-2 infected cells. Emerging Microbes & Infections 2020 9;1 1748-1760 [PMID: 32691695]